Cobalt Chromium Poisoning from Metal on Metal Hip Replacements
Cobalt-Chromium Metal Poisoning from Hip Replacements
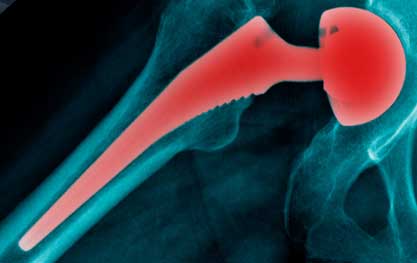
Cobalt-Chromium poisoning is one of many risks for anyone with a metal-on-metal (MoM) hip replacement. It happens when there’s friction between the metal surfaces when the ball and cup of the hip rub together. The friction releases tiny metal particles into your hip joint. The larger particles stay around the hip joint, but the smaller particles can get swept in your bloodstream and travel to other parts of your body.
Your doctor will probably order blood tests to check the levels of metal in your bloodstream. Some tests check the level of metal in your urine instead.
What is Considered a High Level of Chromium and Cobalt?
Slightly elevated metal levels are normal for patients with metal on metal hip replacements. Really high metal levels are alarming. DePuy Orthopedics, Inc. released a report saying that concentrations higher than 7 parts per billion (ppb) of cobalt and/or chromium are of concern. The Mayo Clinic has set a much lower reference value for blood testing for chromium and cobalt, listed below.
High Chromium Levels: Greater than 1ng/mL
According to the Mayo Clinic Medical Laboratories, “blood serum concentrations greater than 1ng/mL in a patient with Cr-based implant suggest significant prosthesis wear.” Their research also indicates that these levels increase the longer you have the hip implant.
High Cobalt Levels: Greater than 10ng/mL
Additionally, the Mayo Clinic Medical Laboratories reports that “cobalt is not highly toxic, but large doses will produce adverse clinical manifestations. Toxic concentrations are greater than or equal to 5.0 ng/mL. Serum concentrations greater than 10ng/mL in a patient with cobalt-based implant suggest significant prosthesis wear.”
What Should I Do if I Have Elevated Metal Levels?
See your orthopedic surgeon and primary care physician immediately. This is a situation that requires long-term medical monitoring.
If your concentration of cobalt and chromium remains above a safe level, your doctor will probably recommend a MARS MRI and/or ultrasound and more testing.
If not, you may want to request these advanced tests from your doctor, even if you don’t have any symptoms.
What if I Don’t Have any Symptoms?
Often there are no immediate physical signs of a problem, but the hidden damage that these metals can do to your body is traumatic. The earlier you get medical care, the better.
Some adverse reactions can be cured by a revision surgery or other medical interventions, but only if done early enough.
This is why it’s important to know what kind metal on metal hip replacement you have, and to ask your doctor if you should get regular blood tests for metal ion levels.
Why is Blood Testing Critical?
Cobalt and chromium blood testing is critical, even if you don’t have any symptoms or physical issues with your hip. You can still have metal poisoning with no symptoms.
Leading orthopedic surgeons recommend cobalt and chromium blood testing every three months for as long as a patient has a metal on metal hip in their bodies.

Need Metal Blood Testing?
Some labs perform these screenings without doctor’s orders
FREQUENTLY ASKED QUESTIONS ABOUT COBALT CHROMIUM POISONING:
What is Cobalt Chromium Poisoning from a Hip Replacement?
Cobalt chromium poisoning happens when the metal parts of a hip implant rub together and release tiny particles of metal in the body. These small metal ions travel through your bloodstream and can damage other parts of your body, such as your kidneys, vision, hearing, and even memory.
How Common is Cobalt Chromium Poisoning from a Hip Replacement?
Cobalt Chromium poisoning is most common in people with a metal on metal hip implant although it can happen in other types of hip replacements. Metal poisoning can be very serious and can lead to many health problems, including metallosis, pseudotumors, tissue and muscle damage, hearing and vision loss, and even memory issues.
How Do You Know if You Have High Levels of Cobalt Chromium in Your Blood?
The only way to find out if you have high cobalt chromium levels is to have your doctor perform blood tests to check the levels of metal in your blood. It’s important to know what kind of hip replacement you have, and to tell your doctor, so that you can receive the proper tests.
What are the Symptoms of Cobalt Chromium Poisoning from a Hip Replacement?
Some people with cobalt chromium poisoning may have no symptoms, which is why it’s important to get your blood levels checked for toxic levels of metal. Symptoms can include:
- Metallosis
- Pseudotumors
- Osteolysis
- ALVAL- Aseptic Lymphocyte Dominant Vasculitis Associated Lesions
- ALTR – Adverse Local Tissue Reaction
- Adverse Reaction to Metal Debris
- Neurological problems
- Vision loss
- Heart problems
- Hearing loss
How is Cobalt Chromium Poisoning from a Hip Replacement Treated? Revision Hip Surgery.
Revision surgery can be needed to fix the injuries caused by defective hip replacements. It involves removing the damaged implant and replacing it with a new one. Many times, a surgeon must repair extensive tissue, muscle, and bone damage from the defective device before the new hip replacement can be put in place. Because of this, revision surgery is a complex operation and requires a specialized orthopedic surgeon to perform it.
Mctlaw Attorneys are Highly Experienced in Metal Hip Lawsuits
- In 2008 mctlaw led the first lawsuit in the United States against a manufacturer of the current generation of defective metal-on-metal hip replacements.
- Mctlaw has reviewed the millions of discovery documents involved in this litigation.
- Other law firms contact our attorneys for advice on these types of cases.
- Our attorneys have personally questioned under oath dozens of orthopedic hip manufacturer’s employees, including surgeons, designers, marketing executives, and corporate CEOs.
- Our attorneys are aggressively and actively litigating defective metal on metal hip cases through the U.S. state and federal court systems.
- We are not waiting for a settlement that may never come because our clients need help now.

Find out now if you have a defective metal hip injury claim
Your case review is free. Find out how to preserve evidence in your case.
Common Adverse Reactions to Metal Ions from Metal on Metal Hip Replacements
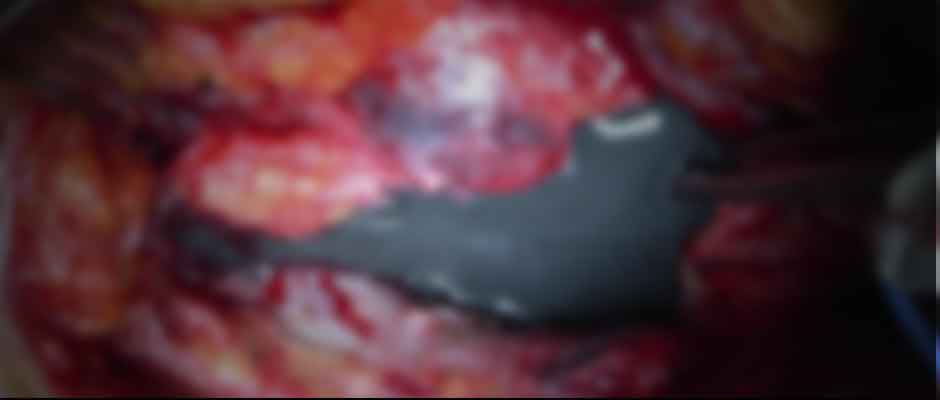
Metallosis Reaction During Hip Revision Surgery
Sometimes the best way to understand severe metal reactions is to see for yourself. The image shows a still shot of metallic-colored fluid gushing from the surgical area near the hip implant.
In this surgery, the patient is a 70-year-old man who reported swelling in his hip. During the revision surgery, doctors punctured the fluid-filled tissue surrounding the muscles of the hip to find a surprising amount of discolored fluid, along with corrosion at the head-neck junction of the hip implant. The pathology report confirmed it was an adverse local tissue reaction (ALTR) from the metal-on-metal hip implant.
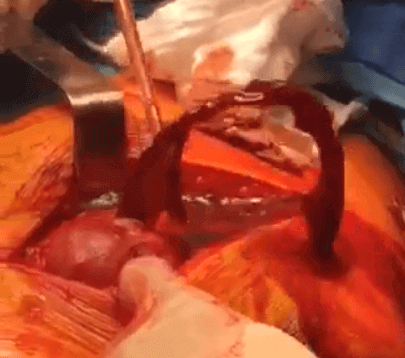
Adverse Reaction to Metal Debris (ARMD)
ARMD describes general complications to metal debris in the body from corrosion in a metal on metal (MoM) hip implant.
The researcher who coined the term “ARMD” used it to describe all the different types of soft tissue damage found in patients with MoM hips, including metallosis, pseudotumors, and ALVAL.
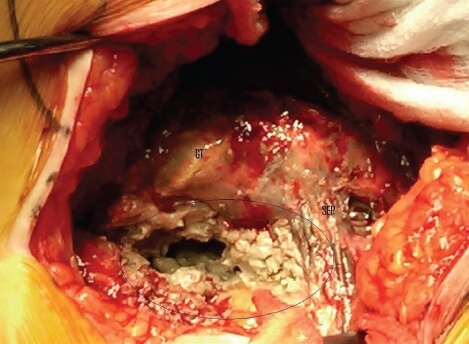
Aseptic Lymphocyte-Dominant Vasculitis-Associated Lesions (ALVAL)
ALVAL happens when the metal particles from the defective metal implant interact with the body’s immune cells, causing the body’s defense system to act like it’s being attacked.
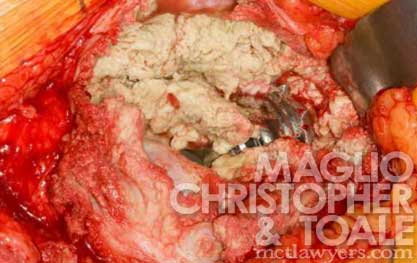
Metallosis
Metallosis is a type of metal poisoning. It involves a build-up of metal debris and particles in the body’s soft tissue.
These particles rot healthy red tissue and muscle around the implant and turn it into a dry, gray, and dead mass of gunk.
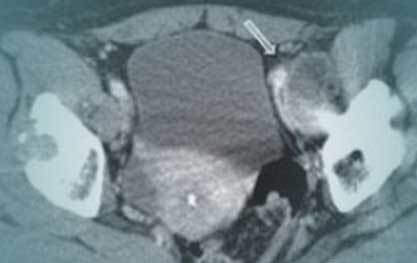
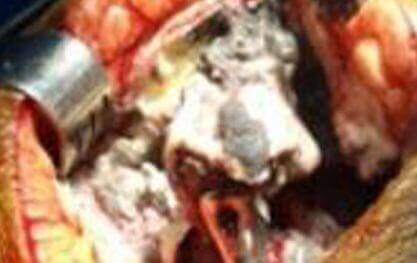
Osteolysis
Bone loss is the destruction of bone around an implant. Bone loss is also called “osteolysis” or “aseptic osteolysis.” It is a common yet serious side-effect of Metal on Metal (MoM) hip replacements.
Adverse Local Tissue Reaction (ALTR)
ALTR stands for “Adverse Local Tissue Reaction,” and it’s a type of inflammatory response, similar to an allergy, that can happen in the hip joints of people with Metal-on-Metal (MoM) hip implants.
Metal on Metal Hip Replacement Cases Across the United States
Click on each state to view the number of cases.
Content Reviewed by Michael Cowgill – Product Liability Lawyer

Michael Cowgill is an experienced attorney in the product liability division at mctlaw. Michael focuses his practice on defective medical devices such as recalled metal-on-metal hips and wrongful death lawsuits involving Kratom. Mr. Cowgill graduated Magna Cum Laude from Lewis & Clark Law School in Portland, OR. He volunteers as a high school mentor with a program for underprivileged youths interested in pursuing a future legal career.
This page was last updated:


Find out right now if you have a claim
Your case review is free. Don’t wait to get help because it could hurt your case.

As an experienced leader in these types of lawsuits, we were confident the firm would have the expertise. However, what surprised us most was the high level of excellent customer service from the firm’s staff!
Pat R.
I can’t recommend this firm enough. They have an outstanding team that truly care for their clients…I have been awarded a fair six figure settlement.
Nate M.
When I say “they went to bat” for me…this Law Firm literally did just that. They persevered to bring the hard-nosed Manufacturer to settle and provide me some recompense for everything I had to endure which led to this suit.
Me’Chelle




