Heart Failure or Cardiomyopathy from Metal on Metal Hips
Heart Failure and Cardiac Problems from Metal on Metal Hips
There is strong medical evidence that metal on metal hip replacements sometimes cause heart failure and other life-threatening cardiac problems. These types of reactions are known as “systemic issues” because they trigger a reaction in organs throughout the body, not just around the hip socket.
The US Food and Drug Administration (FDA) issued a serious warning to the public about heart problems linked to metal on metal hips, including heart attacks. To read the warning, click on the image.
When a patient moves, the metal surfaces on the cup and ball of the hip implant rub together, consequently releasing tiny microscopic particles of cobalt and chromium metal into the bloodstream. This is called metallosis, or cobalt poisoning. The increased levels of cobalt and chromium flow through the blood and into other organs in the body such as the heart, causing severe reactions like cobalt cardiomyopathy and heart attack.
Making a Legal Case for Metal on Metal Hip Replacement and Cardiac Problems
As attorneys litigating these cases, we strongly suspect cardiac problems can be caused by the toxic metals released from these hip implants. You should tell your doctor if you have a metal on metal hip and are having heart problems. Often these symptoms are warnings that something is wrong with the hip implant. If your doctor is not aware that you have a metal on metal hip, they may decide the diagnosis is unrelated to the toxic metal poisoning.
The science that links systemic issues like heart damage to metal on metal hips is not yet fully recognized in the medical community, but can be seen as evidence of a larger problem with a hip implant. The connection between metal on metal hips and high metal levels, pseudotumors, and metallosis is well documented.

Find out now if you have a defective metal hip injury claim
Your case review is free. Find out how to preserve evidence in your case.
Report Your Problems to the FDA
There is something you can do: Report your adverse metal on metal hip experience to the FDA by filling out a MedWatch form.
Hip manufacturers are required to report all hip failures to the FDA but often find loopholes to keep this data hidden.
If the FDA doesn’t have accurate reporting of patients suffering from serious complications, neither does your surgeon. Your doctors rely on FDA data to diagnose problems and know when a specific type of hip implant is having issues.
Don’t let the hip implant makers brush your information under the rug. Most people don’t know that you can self-report problems. However, the FDA needs to hear from you. Empower yourself against these huge corporations that put profits above your health.
It’s simple to fill out and an important way to make your voice heard. Here is a link to the form:
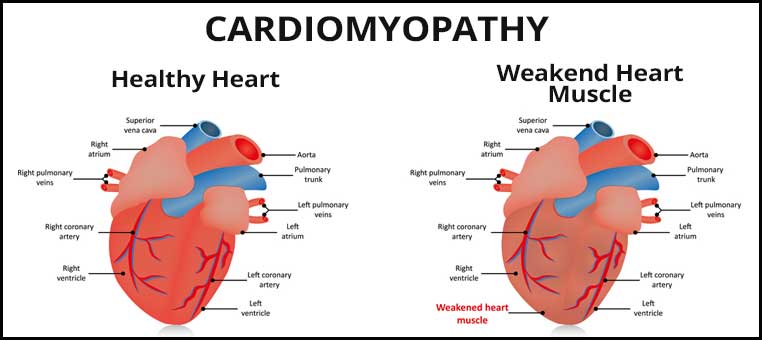
Cobalt Cardiomyopathy from Metal on Metal Hip Implants
Cobalt cardiomyopathy is a thickening of the heart muscle. Unfortunately, this thickening makes it difficult for the heart to contract and pump blood through the body. Cardiomyopathy gets very severe very quickly, leading to complete heart failure or heart attack in a very short amount of time.
Signs and symptoms that you may have cobalt cardiomyopathy:
- Chest pressure
- Chest pain
- Difficulty breathing
- Excessive fatigue and sweating during regular daily activity
- Loss of some feeling in the feet
- Kidney failure
- Loss of hearing
- Loss of vision
- Weight loss
- Lethargy
- Tachycardia
What Cobalt Cardiomyopathy Can do to the Heart:
High levels of cobalt in the heart can have many negative effects on the heart’s function. If you have cobalt cardiomyopathy, your physician may tell you that you are experiencing some of the following problems:
- Biventricular dysfunction: Failing of both heart ventricles.
- Myocyte hypertrophy: Enlarged heart muscle cells.
- interstitial fibrosis: Inflammation and scarring of the heart tissue.
- Congestive heart failure (creating a need for transplant or LVAD implantation to avoid death).
- Lowered left ventricular ejection fraction: Slowed blood-pumping ability in the left ventricle.
- Pericardial effusion: Abnormal accumulation of fluid around the heart.
- Mitral regurgitation: A leakage of blood in one of the heart valves.
- diastolic dysfunction: A type of heart failure where blood collects in the lower half of the heart.
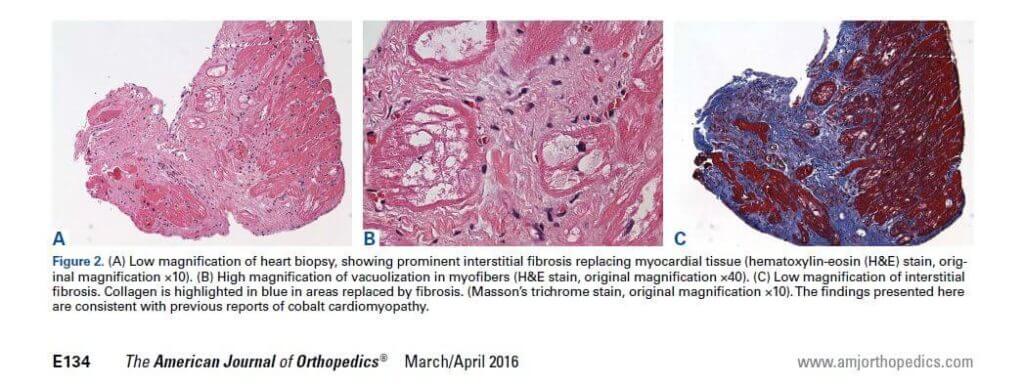
Diagnosis
If you are exhibiting symptoms of cobalt cardiomyopathy, your doctors should conduct an MRI scan of your heart and/or an ultrasound of your heart (echocardiogram) in order to assess your heart function. Diagnosis of cobalt cardiomyopathy may also require testing tissue samples from the heart, testing cobalt levels in the blood, and evaluating your symptoms.
Does Cobalt Cardiomyopathy Have a Cure?
Often, revision surgery on the hip implant can reverse cobalt cardiomyopathy. However, in some cases, if heart failure is too severe, the patient’s health can continue to deteriorate even after revising the implant and reducing cobalt levels.
Unfortunately, if left untreated or caught too late, cobalt cardiomyopathy can get worse very quickly. Worst case scenarios for cobalt cardiomyopathy include complete heart failure and death.
Content Reviewed by Michele Stephan – Product Liability Lawyer

Michele Stephan handles product liability cases in state and federal courts throughout the country. Ms. Stephan has represented clients in civil litigation for over 25 years. At mctlaw she litigates cases for clients with defective hip replacements and defective drug and pharmaceutical lawsuits. Ms. Stephan is a member of the Twelfth Judicial Circuit Pro Bono Committee and a Director of the Sarasota County Bar Association.
This page was last updated:


Find out right now if you have a claim
Your case review is free. Don’t wait to get help because it could hurt your case.

As an experienced leader in these types of lawsuits, we were confident the firm would have the expertise. However, what surprised us most was the high level of excellent customer service from the firm’s staff!
Pat R.
I can’t recommend this firm enough. They have an outstanding team that truly care for their clients…I have been awarded a fair six figure settlement.
Nate M.
When I say “they went to bat” for me…this Law Firm literally did just that. They persevered to bring the hard-nosed Manufacturer to settle and provide me some recompense for everything I had to endure which led to this suit.
Me’Chelle