Inspire Sleep Apnea Recall
If your Inspire sleep apnea implant has caused you pain or required a revision surgery, you are facing a situation no patient should have to endure. Whether you have experienced “zapping” sensations, inconsistent therapy, or have been told your device is defective, mctlaw’s product recall attorneys are here to help.
We are actively investigating cases involving Inspire Implantable Pulse Generators (IPGs). Although the FDA issued its most serious (Class 1) recall in June 2024 for Models 3024 and 3028, we are hearing from patients with various models who are suffering from similar defects. Our goal is to hold the manufacturer accountable for the safety of these devices.
IMPORTANT:
Contact us now for an Inspire Sleep Apnea Case Investigation.
There is no cost, no pressure, and no obligation
What Is the Inspire Implant for Sleep Apnea?
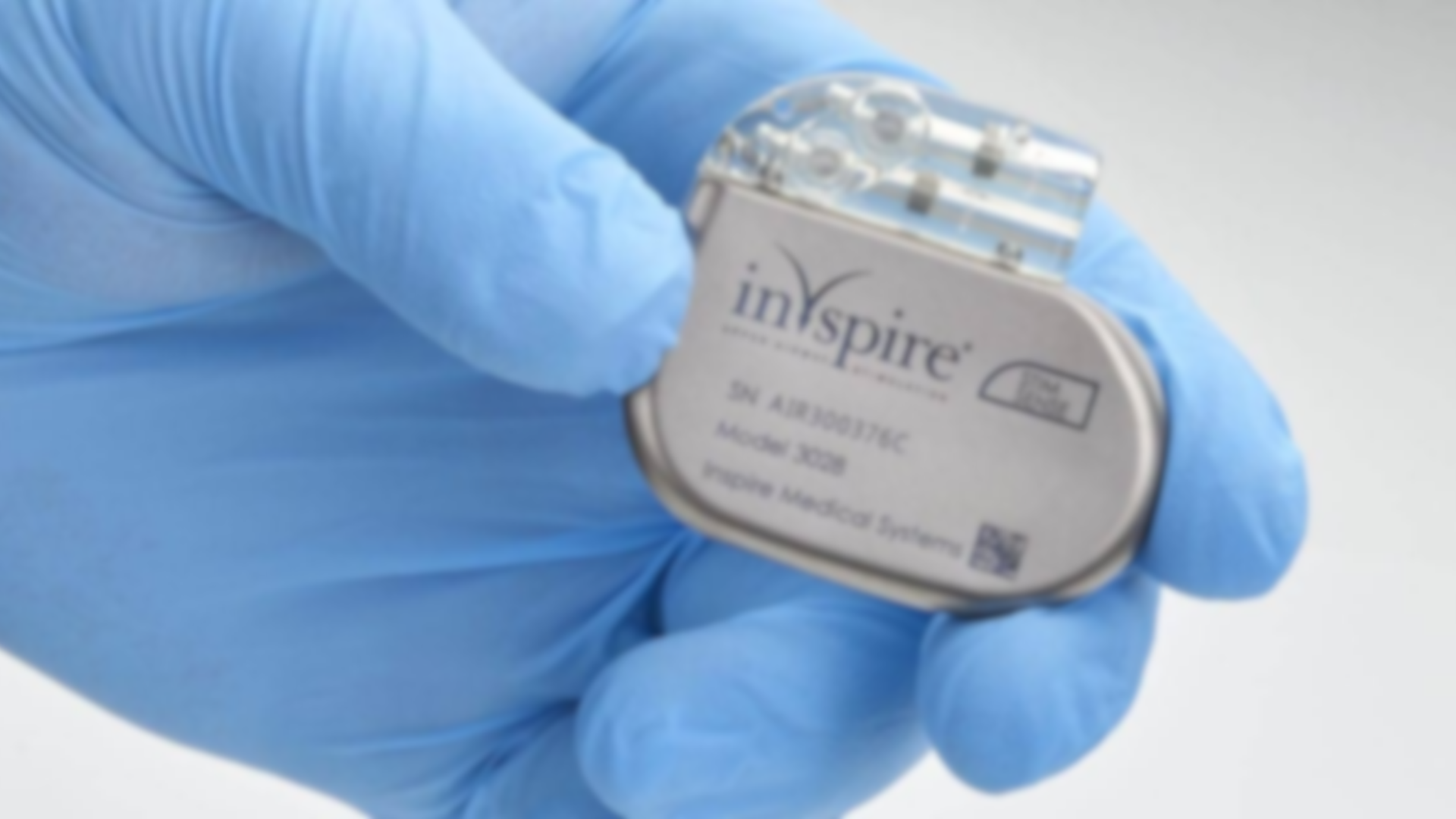
The Inspire implant is a surgically placed device designed to treat obstructive sleep apnea (OSA), a condition that causes repeated breathing interruptions during sleep. OSA happens when soft tissue in the throat collapses, partially blocking the airway. While many patients rely on CPAP machines, those who cannot tolerate them often turn to Inspire as an alternative. The Inspire implant is implanted near the collarbone and works by sending mild electrical pulses to the airway muscles. It uses mild electrical stimulation that sends pulses to the airway muscles, moving the tongue out of the way so users can breathe.
Despite its promise of a better night’s sleep, certain models of the Inspire system have failed to meet safety standards. In June 2024, the FDA issued a Class 1 recall, its most serious type, for two Inspire IPG models: 3028 and 3024. The recall warns that these sleep apnea devices may cause electrical leakage or deliver painful shocks due to a manufacturing defect that can lead to electrical leakage.
Do I Qualify for an Inspire Case Investigation?
We are currently evaluating legal claims for individuals who have experienced complications with the Inspire sleep apnea system. You may qualify for an investigation if you meet any of the following criteria:
- You are experiencing electrical shocks or jolts from the device while it is turned off.
- Your Inspire IPG has been removed or revised due to unintended electrical shocks while it is turned off or because of the product recall.
- You have revision surgery scheduled, or your doctor has recommended removal of the device due to issues with unintended electrical stimulation.
It is important to note that you do not need to have one of the officially recalled model numbers to be considered for our investigation. We are reviewing reports from all patients experiencing abnormal electrical behavior or device failure, as current evidence suggests these issues may extend beyond the models listed in the 2024 recall.
Reports of Health Risks with Inspire Medical IPG Devices
Many affected patients have reported serious complications with their Inspire medical devices, including unexpected electrical shocks even when their Inspire device is turned off. In many cases, people say their concerns were dismissed by healthcare providers or device representatives. Our goal is to make sure these experiences are validated, documented, and evaluated so the right people can be held accountable.
Some patients describe these kinds of unusual complications with their Inspire implants:
- Perceived shocking sensations affecting the tongue muscles or throat
- A complete stop in therapy due to battery failure
- Frustration after doctors or company reps said their Inspire medical device was “working normally”
If you have experienced symptoms such as fatigue, daytime sleepiness, or recurring snoring, you should contact your physician for a thorough evaluation.Even if your Inspire model was not recalled, we recommend you to speak to our attorneys who are actively investigating cases involving Inspire implant electrical stimulation problems or device failures. Please note that we are currently only able to take lawsuits where the patient experienced shocks when the device was turned off.

Inspire Sleep Apnea Recall: What You Need to Know
We want to be upfront: Inspire sleep apnea recall lawsuit is an ongoing investigation. Based on the product recall and reports from patients, we believe there is enough concern to take a closer look. While we may accept your case for a thorough investigation, this phase requires a detailed review of your medical records and any documentation regarding your specific Inspire model and the symptoms you are experiencing.
As we gather more information, we may determine that your situation does not align with the specific legal claims our firm is currently prepared to pursue. Because this investigation is in its early stages, there is no guarantee that your case will proceed to litigation or result in a financial recovery. Our decision to move forward with legal action will depend entirely on the findings of our initial review of your circumstances.
In the meantime, your health and safety are the most important priorities. Even as we investigate the legal aspects of this situation, there are several steps you should take to manage your medical care and protect your well-being:
- Contact the doctor who manages your Inspire device as soon as possible to schedule a routine office visit and rule out any issues
- Pay close attention to how you feel each day – record any unusual physical sensations, such as unexpected zapping or shocks, even if they occur while the device is supposedly turned off
- Keep a personal log of any malfunctions or symptoms you experience, including the date and a description of what happened
Most importantly, you should not stop using your device or attempt to adjust the settings yourself without first speaking to your sleep physician. They can determine if your specific unit is affected and decide on the safest course of action for your respiratory health.
Inspire Sleep Apnea Recall: FAQs
Which Inspire Models Are Actually Under Recall?
In June 2024, the FDA issued a Class 1 recall for the Inspire IV Implantable Pulse Generator (IPG) Models 3028 and 3024.
What Are the Warning Signs that My Device Might Be Failing?
It is important to recognize the warning signs of a failing Inspire implant. Many patients with defective units often report sharp, painful jolts even while the device is turned off. You might also notice a sudden return of sleep apnea symptoms, such as loud snoring, or daytime exhaustion, indicating the therapy is no longer effective. Other red flags include tenderness or swelling around the implant site or neck. Often, these issues are first caught during routine medical check-ups when a doctor detects internal electrical leaks or waveform irregularities.
Can I Still Be Part of the Investigation If My Model Wasn’t Recalled?
Yes. Many sleep apnea patients have experienced shocks when the device is turned off. If you have experienced such issues, please contact us today.
Does the Inspire V Have the Same Problems?
While the Inspire V was marketed as a next-generation solution, it has been the subject of significant legal and commercial scrutiny in 2025 and early 2026. However, the investigation is still ongoing, so it is too early to give any conclusion.
Was Your Inspire Device Affected? Contact mctlaw’s Defective Medical Device Recall Attorneys
Reach out to us regarding the Inspire device recall with the confidence that your consultation is confidential and carries no obligation to file a lawsuit. Speaking with our team is simply the first step in helping us understand what is happening to patients such as yourself. Your experience with an affected product could be key in determining whether this situation leads to broader legal action.
The attorneys at mctlaw have decades of experience handling defective medical device cases across the country, including and especially those that result in serious adverse health consequences. Whether you are struggling with unexpected shocks, unusual sensations, or any other concerns regarding your Inspire system, we are here to listen and help you navigate the road ahead.Medical device recall attorneys Jessica Olins, Altom Maglio, and Michael Cowgill are leading this investigation. We are here to help you understand your options. Contact us for a free consultation.
Content Reviewed by Altom Maglio – Attorney

Altom M. Maglio represents individuals across the United States in complex civil litigation, including COVID eviction moratorium takings claims, rails to trails conversions, listeria outbreak lawsuits, defective orthopedic devices and product liability lawsuits. Mr. Maglio founded mctlaw in 1999 to represent people in their fight against huge corporations and large government programs. He is also an active leader in the U.S. Court of Federal Claims Bar Association and the American Association for Justice Leadership Forum.
This page was last updated on:


Find out right now if you have a vaccine injury claim
Your Case Review is Free. Don’t Wait to Get Help Because There is a Deadline.
The COVID-19 Vaccine is NOT Eligible

As an experienced leader in these types of lawsuits, we were confident the firm would have the expertise. However, what surprised us most was the high level of excellent customer service from the firm’s staff!
Pat R.
I can’t recommend this firm enough. They have an outstanding team that truly care for their clients…I have been awarded a fair six figure settlement.
Nate M.
When I say “they went to bat” for me…this Law Firm literally did just that. They persevered to bring the hard-nosed Manufacturer to settle and provide me some recompense for everything I had to endure which led to this suit.
Me’Chelle